This study used data from the Swedish CArdioPulmonary bioImage Study (SCAPIS)14, a Swedish nationwide population-based cohort mainly designed to research cardiovascular and chronic obstructive pulmonary diseases. All Swedish residents have a unique personal identification number, which allowed unbiased and randomized recruitment from the Swedish population register. Initial contact was made by sending out an informational brochure asking recipient to contact the study centre. If the centre was not contacted, the recipient was reminded by up to three telephone calls and finally by letter. If the centre was contacted and the recipient was willing to participate in the study, an appointment was arranged at the study centre. No exclusion criteria applied except the inability to understand written and spoken Swedish for informed consent.
A protocol of the statistical analysis plan, was registered on the open science framework before start of the analysis15. The Regional Ethical Review Board approved SCAPIS as a multicentre trial (2014-33-32 M). All participants provided written informed consent, and the research was conducted in accordance with the Declaration of Helsinki. This study received ethical approval on 2021-05-18 from the Swedish Ethical Review Authority (Dnr 2021-02,121).
Outcomes and measures
Participants of SCAPIS were asked to complete a questionnaire, comprising of 140 questions, including questions on biological sex, age, socioeconomic factors, social support, history of disease, family history of disease, alcohol consumption, smoking, physical inactivity, and intake of food and drinks.
Socioeconomic factors assessed included educational level, employment status, marital status, financial preparedness for unforeseen expenses, budget management difficulties, living situation, and own and parents’ country of birth. Social support was evaluated using a validated condensed version of the Interview Schedule for Social Interaction16, with each of the twelve items assessed separately. Analysing each item separately allowed us to study which parts of social support specifically is associated with health behaviours, rather than only looking at social support as a whole, summarised into a single score. Self-reported medical history encompassed various conditions, including heart-related conditions (e.g. myocardial infarction and heart failure), lung diseases (e.g. chronic obstructive pulmonary disease (COPD) and chronic bronchitis), diabetes, and cancers. Family history of diseases in first-degree relatives was also queried.
Unhealthy alcohol consumption was assessed using the alcohol use disorders identification test (AUDIT)17. In this study, the last two questions (“Have you or someone else been injured because of your drinking?” and “Has a relative, friend, doctor, or other health care worker been concerned about your drinking or suggested you cut down?”) had only two response options (No/Yes) instead of the usual three (No/Yes, but not in the last year/Yes, during the last year). The AUDIT score ranges from 0 to 40, with higher scores indicating more harmful alcohol use. A score of 8 or higher typically indicates hazardous or harmful alcohol consumption. The Cronbach’s alpha in this study was 0.73.
Nonadherence to dietary recommendations was determined through food intake assessments using the MiniMeal-Q questionnaire18. This questionnaire covered 75 to 126 food items and inquired about dietary habits over the past few months. Energy (kJ/day) and macronutrient (g/day) intake were calculated using the national nutrient content database (www.slv.se). The Swedish Healthy Eating Index score (SHEI-score) was utilised to gauge adherence to dietary recommendations19. This index reflects the ratio between the recommended consumption of nine specified foods and the nutrient intake outlined in the 2012 Nordic Recommendations. These nine specified foods and the nutrient intake were:
-
1.
At least 500 g vegetables and fruit per day (potatoes not included)
-
2.
At least 2.5 g fibre/MJ
-
3.
At least 75 g wholemeal/10 MJ
-
4.
At least 45 g fish and shellfish per day (frequency of 2–3 per week and portion size 125 g)
-
5.
Polyunsaturated Fatty Acids (PUFA) minimum 7.5 E%
-
6.
Monounsaturated fatty acids (MUFA) minimum 15 E%
-
7.
Saturated Fatty Acids (SFA) maximum 10 E%
-
8.
Maximum 500 g red and processed meat per week
-
9.
Maximum 10 E% added sugar
Scores for each item range from 0 (nonadherence to the dietary recommendation) to 1 (adherent to the dietary recommendation), and the total score ranges from 0 to 9, with lower scores indicating poorer adherence to dietary recommendations. It’s worth noting that, for consistency with other behaviour measures, the scale was reversed in the analyses enclosed, so higher scores indicate lower adherence and vice versa. Participants in the first, second, and third quartiles were considered non-adherent to dietary guidelines.
Physical inactivity was defined following the definition by the World Health Organization as insufficient physical activity to meet current recommendations20. For adults aged 18–64, this entails at least 150–300 min of moderate-intensity aerobic activity or at least 75–150 min of vigorous-intensity aerobic activity per week. It should also include muscle-strengthening activities involving all major muscle groups two or more days per week. Physical inactivity was assessed with a question regarding exercise frequency over the past three months to improve endurance and well-being. Response options included: Never; Sometimes, but not regularly; 1–2 times a week; 2–3 times a week; More than 3 times a week; Not willing/able to reply. “Never” or “Sometimes, but not regularly” indicated physical inactivity as these participants were considered to not have sufficient physical activity to meet current recommendations.
Smoking status was determined with a single question: “Do you smoke?” Response options included: No, I never smoked; No, I quit smoking; Yes, I smoke occasionally; Yes, I smoke regularly; Not willing/able to reply. Participants who stated they were current smokers during an oral inquiry at a study visit, but answered on the questionnaire that they did not smoke, were considered current smokers.
Data analyses
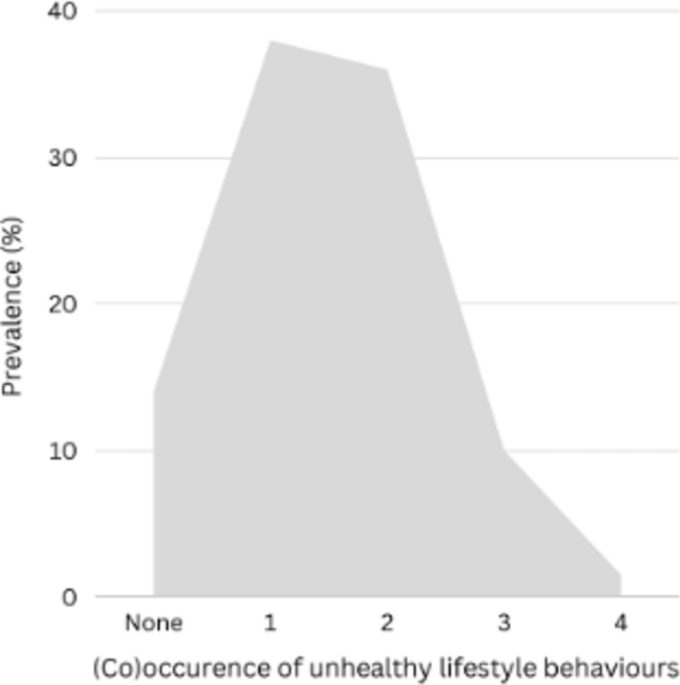
We used multilevel multinomial regression to estimate the cohort prevalence of co-occurrence of two and three unhealthy lifestyle behaviours and the prevalence of all four unhealthy lifestyle behaviours. The levels were the six different study centres for which adaptive intercepts were added to the models. ‘No unhealthy behaviours’ was used as the reference category, and adaptive intercepts were added for study site. We added covariates to the multinomial regression models to estimate conditional associations between the co-occurrence of unhealthy lifestyle behaviours and sociodemographic characteristics (including age and biological sex), socioeconomic factors, social support, and history of disease. To estimate the associations among unhealthy lifestyle behaviours, we regressed each health behaviour measure (AUDIT, SHEI-score, physical inactivity, and smoking) against the other health behaviour measures, e.g., for AUDIT, we used SHEI-score, physical inactivity, and smoking as covariates. Interactions among the covariates were also added. We used negative binomial regression for AUDIT, linear regression for SHEI-scores, and logistic regression for physical inactivity and smoking. These models also included adaptive intercepts for study site.
We used Bayesian inference to estimate posterior distributions of associations21. Unlike maximum likelihood inference with null hypothesis testing, Bayesian inference results in a probability distribution over quantities of interest (in this case, associations). Thus, rather than focusing only on rejecting or keeping the narrow null hypothesis, Bayesian inference assigns a probability to each possible value of the associative measure (e.g. an odds ratio). This allows for a more straightforward inspection of the relative compatibility between the data and different association estimates. The distribution of estimates is referred to as the posterior distribution since it incorporates our prior belief (prior distribution) and the data observed. For reporting it is convenient to communicate a point estimate of the association rather than the full posterior distribution, and we used the median of the posterior distribution for this purpose. To portray the posterior distribution and the inherent uncertainty of the estimates, we used 95% compatibility intervals (CI) defined as the 2.5% and 97.5% percentiles of the posterior distributions. We used standard normal priors for all parameters in the multinomial cohort prevalence models. In models with covariates, we used Cauchy priors to induce sparsity, with a half-normal hyperprior for the Cauchy scale parameter. Data were analysed using the R statistical software version 4.0.4 and Stan 2.30.1 (CmdStan).