Study population
Analyses were performed in the NHS and HPFS. The NHS was established in 1976 and included 121,700 US female registered nurses aged 30–55 years33. The HPFS was initiated in 1986 and recruited 51,525 male health professionals aged 40–75 years34. Details about the cohorts were published previously33,34. Biennial assessments of lifestyle factors and medical history were conducted through mailed questionnaires, maintaining a follow-up rate exceeding 90%. High validity and reproducibility of self-reported health information in the cohorts has been reported previously35,36,37. For both cohorts, the baseline for this analysis was 1986, that is, when the food frequency questionnaires (FFQ) allowed for computing all dietary pattern scores.
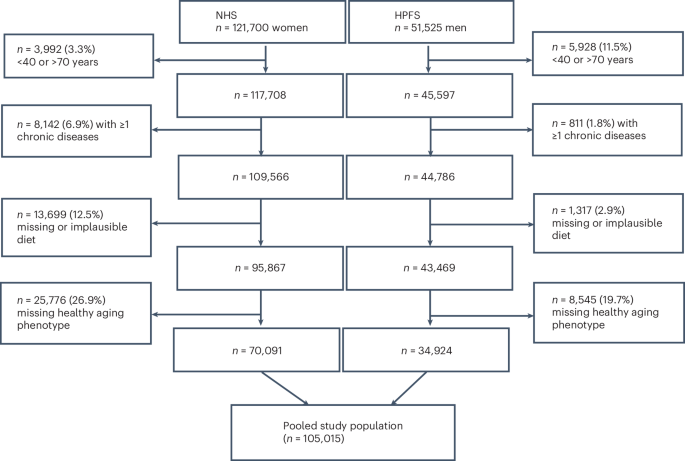
Figure 1 shows the flow diagram of the study. Participants included in this analysis had answered the 1986 questionnaires and had a chance of attaining 70 years at the end of the follow-up (aged between 39 and 69 years at baseline; n = 9,920 excluded). Participants with a history of one or more of 11 major chronic diseases, including cancer (except for non-melanoma skin cancers), diabetes, myocardial infarction, coronary artery disease, congestive heart failure, stroke, kidney failure, chronic obstructive pulmonary disease, Parkinson disease, multiple sclerosis and amyotrophic lateral sclerosis, at baseline (n = 8,953), with missing data for one or more dietary pattern scores between 1986 and 2010, or who reported implausible total energy intakes (<500 or >3,500 kcal d−1 for women and <800 or >4,200 kcal d−1 for men) (n = 15,016) and with missing healthy aging phenotype (n = 34,321) were excluded.
The study protocol was approved by the institutional review boards (IRBs) of the Brigham and Women’s Hospital (BWH), Harvard T.H. Chan School of Public Health (HSPH) and participating registries (IRB protocol nos. 2001P001945/BWH and 10372) as required. Per long-standing IRB-approved procedures at BWH and HSPH, the completion and return of the baseline and subsequent follow-up questionnaires implies consent. Before 2006, consenting practices varied and many collections were conducted with implied consent. A waiver of documentation for informed consent was granted for these studies. Written informed consent was required for medical record acquisition.
Dietary assessment
Dietary intake was measured using a validated FFQ with more than 130 items administered in 1986 and every 4 years thereafter. The validity and reliability of the FFQ for measuring nutrient, food and food group intakes have been reported previously38,39. Participants were asked how frequently they consumed specific foods in the past 12 months. Nutrients were calculated using the United States Department of Agriculture (USDA) and Harvard University Food Composition Databases40, and supplemented with biochemical analyses. Based on the reported nutrient and food intakes, eight dietary pattern scores were computed, including the AHEI, aMED, DASH, MIND, hPDI, PHDI, EDIP and EDIH, as well as UPF consumption. The AHEI, aMED, DASH, MIND, hPDI, and PHDI scores, and UPF consumption, have been developed based on dietary recommendations and available evidence. The scores showed moderate-to-high validity and reproducibility41 and were associated with risk of health outcomes28,42,43,44. The EDIP and EDIH were empirically derived using biomarkers of chronic inflammation45 and hyperinsulinemia46, respectively.
The AHEI score was computed based on the intake of 11 foods and nutrients that have been positively or inversely associated with chronic disease risk, including fruits, vegetables, nuts and legumes, alcohol, red and processed meat, whole grains, sodium, trans fats and long-chain n-3 (omega-3) fatty acids47. Each component was scored on a scale from 0 to 10 and the AHEI ranged from 0 to 110, a higher score indicating better overall diet quality. Adherence to the Mediterranean diet was assessed using a modified version of the 9-point aMED score21. The score was calculated based on adherence to nine food items (with either higher or lower intake) that characterize the Mediterranean diet: vegetables; legumes; fruits; nuts; whole grains; fish; moderate alcohol consumption; lower intake of red and processed meat; and monounsaturated to saturated fatty acids21. The modified aMED score ranged from 0 to 9, a higher score denoting a higher adherence to the Mediterranean diet21. The DASH score, designed to help prevent or treat hypertension, was based on eight dietary factors, including intake of high fruits, vegetables, nuts and legumes, low-fat dairy products, whole grain intake, low sodium, sweetened beverages and processed and unprocessed red meats23. Each component was attributed a score between 0 and 5 based on intake distribution quintiles. The overall score ranged from eight (lowest adherence) to 40 (highest adherence). The MIND score included ten brain-healthy dietary factors, including leafy-green vegetables, other vegetables, berries, nuts, whole grains, fish, beans, poultry, wine (in limited amounts) and olive oil, and five less healthy factors, including butter and margarine, cheese, red meat, fast/fried foods, and pastries and sweets22. Points of 0, 0.5 or 1 were attributed to each and the total MIND score ranged from 0 to 15, a higher score indicating a higher adherence. The hPDI was designed to reflect adherence to a healthy plant-based diet48; it included 18 dietary factors and ranged from 18 (lowest adherence) to 90 (highest adherence). The PHDI was derived based on the adherence to the reference diet included in the EAT-Lancet report49. The reference diet was based on evidence for health outcomes and staying within planetary boundaries for greenhouse gas emissions and other environmental parameters24. The scoring criteria included 15 dietary factors. For each dietary factor, the score ranged from 0 to 10, except for non-soy legumes and soy foods, which contributed a maximum score of 5. The total PHDI score ranged from 0 (non-adherence) to 140 (perfect adherence). The EDIP was scored based on 18 dietary factors related to inflammation50. The EDIH included 18 dietary factors related to hyperinsulinemia46. Given that higher EDIH and EDIP scores denote higher hyperinsulinemia and inflammatory dietary patterns, respectively, the orientation of the scores were reversed to ease comparison with other dietary scores and were denoted as rEDIH and rEDIP. Figure 3 is a visual representation of the dietary patterns and their dietary factors.
Food items were also categorized according to the NOVA classification system51. The average daily energy intake from UPFs was calculated, and the percentage of total daily energy from UPFs was determined based on the average total energy intake.
Assessment of healthy aging
Based on the construct of ‘successful aging’ described by Rowe & Kahn52 and previous reports19,53,54,55,56, a healthy aging phenotype was defined as surviving to the age of 70 years without the presence of 11 major chronic diseases and with no impairment in cognitive function, physical function or mental health. Participants who did not meet the phenotype criteria or who died before 2016 were considered as usual agers. Those who died after 2016, but had a complete assessment of the phenotype were included in the analyses. Cognitive function was assessed using a validated Subjective Cognitive Decline questionnaire57 in which reporting one or fewer of seven (NHS) or six (HPFS) cognitive concerns was considered as intact cognitive function. Physical function was assessed using the 36-item Short Form survey58 subsection on physical limitations that includes ten questions. Participants who reported not being limited in climbing one flight of stairs, walking one block, walking more than a mile, vacuuming, moving a table, walking several blocks, bathing or dressing, and being a little or not limited in climbing several flights of stairs, running lifting, bending, kneeling stooping and lifting carrying groceries, were considered as having intact physical function. Mental health was evaluated using the 15-item Geriatric Depression Scale, which ranges from 0 to 15 with a lower score indicating fewer depressive symptoms59. Reporting 1 or fewer depressive symptom (median value of both cohorts) was considered as having intact mental health. The 11 major chronic diseases were selected as they are the primary causes of mortality in the United States60 or are considered to be highly debilitating. They were also those ascertained at baseline and throughout the follow-ups. Clinical diagnoses of the major chronic diseases were reported by participants or next of kin for the deceased and were then ascertained by a physician through medical records, pathology reports or supplemental questionnaire inquiries, which are highly valid in these cohorts61. In the NHS, cognitive function was assessed in 2014; the presence of chronic diseases, physical function and mental health were assessed in 2016. In the HPFS, all domains were assessed in 2016.
Assessment of covariates
Every 2 years, participants completed a questionnaire that queried on body weight, ancestry, smoking status, physical activity, multivitamin use ever, family history of chronic diseases, including myocardial infarction, type 2 diabetes, cancer and dementia, postmenopausal status and hormone use in women, marital status, living alone and history of depression (based on antidepressant medication use and self-report of depression). The questionnaires are available online at https://nurseshealthstudy.org/participants/questionnaires for the NHS and at https://www.hsph.harvard.edu/hpfs/hpfs-questionnaires/ for the HPFS. BMI was calculated by dividing the weight (kg) by the height (m2). SES was calculated as a composite score using the participant’s geocoded address, combining US census household income, home value, college degree, ancestry, occupied housing units and unemployment, and whether families were receiving interest or dividends62.
Statistical analysis
The follow-up duration was calculated from the date when the first FFQ was returned until the date of the participant’s death or the date of assessment for healthy aging in 2016, whichever occurred first. The main analysis used the average of available dietary pattern scores between 1986 and 2010 as the exposure to capture long-term intake and allow a lag of 6 years between exposures and outcomes. The residual method was used to adjust dietary pattern scores for total energy intake, including energy from alcohol63, and quintiles were calculated. Missing values for covariates were replaced with the cohort-specific median value. As outcomes are binary and measured at one time point, logistic regression models were used to estimate the ORs and 95% CIs of healthy aging and its individual domains according to quintiles of energy-adjusted dietary pattern scores, with age at baseline in 1986 as a covariate (base model). The multivariable-adjusted model was further adjusted for baseline-reported ancestry (European, Asian, African-American, Other), SES (composite score) and marital status (yes/no), ever used multivitamins (yes/no), ever lived alone (yes/no), family history of dementia (yes/no), myocardial infarction, cancer, and diabetes (yes/no), and medical history of depression (yes/no), diabetes and CVD (yes/no), smoking status in 2010 (never, former, current smoker: 1–14 cigarettes per day, 15–24 cigarettes per day and ≥25 cigarettes per day), postmenopausal status (yes/no) and menopausal hormone use (no, past, or current hormone use; women only), and mean (1986–2010) BMI (kg m−2), physical activity (MET-h week−1) and alcohol intake (g per day) for DASH, hPDI and PHDI. An OR above 1 indicated a higher likelihood of healthy aging. Associations using the patterns as continuous variables, standardized according to their respective increment from the 10th to the 90th percentiles, were also examined. Analyses were performed in each cohort and in the pooled cohorts. The analyses in the pooled cohorts were further adjusted for cohort (sex). Absolute risks in both quintiles were calculated using the proportion of healthy agers in Q1 and the multivariable-adjusted OR comparing Q5 versus Q1. E values and the lower or upper bound were calculated for the ORs to assess the potential impact of unmeasured confounding on the observed associations. A higher E value denotes stronger evidence that unmeasured confounding would be required to fully explain away the observed association. Prespecified subgroup analyses were conducted according to cohort (sex), BMI (<25 versus ≥25 kg m−2), physical activity (above/below median), smoking (yes/no), SES (above/below median) and ancestry (European/non-European). Potential subgroup interactions were evaluated using interaction models, including the product of the subgroup and the exposure. As secondary analyses, the association of dietary patterns scores with healthy aging using an age cutoff of 75 years, and the association of UPF consumption with healthy aging (>70 years) were assessed. As a sensitivity analysis, we applied linear weights to dietary pattern scores across seven time points, ranging from 1986 to 2010, assigning the highest weight to the year closest to the healthy aging assessment (2010) and the lowest weight to 1986.
Intake in the individual food groups and nutrients that reflect the dietary pattern scores were also energy-adjusted and standardized according to their respective increment from the 10th to the 90th percentiles. The associations of energy-adjusted food group and nutrient intakes with healthy aging and its domains were examined using logistic regressions, accounting for the covariates described in the multivariable-adjusted model. Given that all dietary factors are also found in the pattern scores, the models were not adjusted for any measure of diet quality. P values were corrected for FDR using the Benjamini–Hochberg method; an FDR-corrected P < 0.05 was considered statistically significant.
Analyses were performed with SAS v.9.4 (SAS Institute) and R v.4.2.0. Statistical tests were two-sided and P < 0.05 was considered statistically significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.